WOUND CARE
Human allograft wound care solutions from MTF Biologics. Dermal matrices, placental membranes, and adipose tissue allografts for acute and chronic wound management.
APMA Seal of Approval — SomaGen Meshed & Leneva
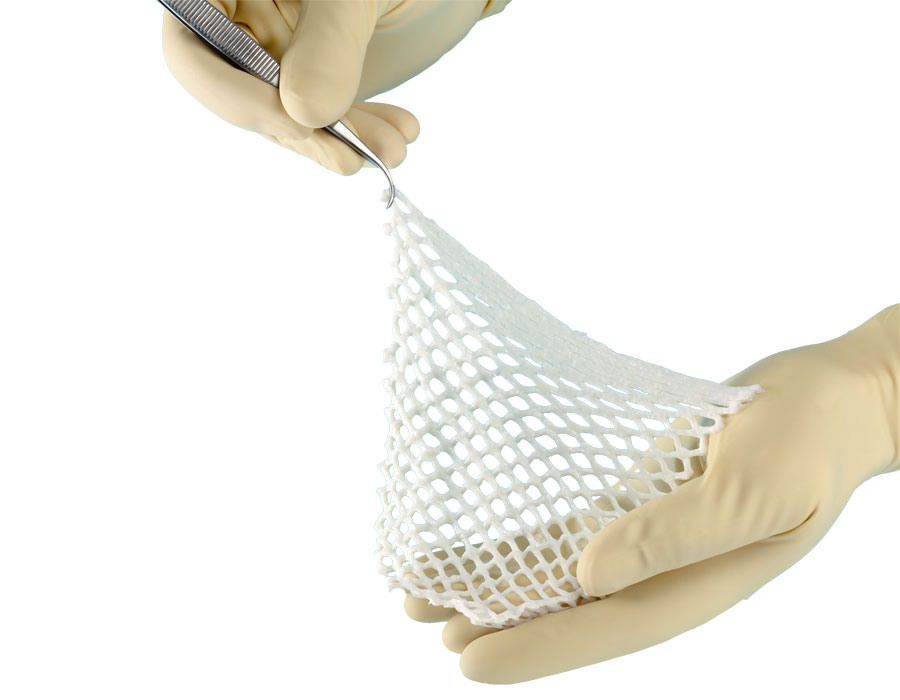

Dermal Matrix
SomaGen® Meshed
Acellular human reticular dermal allograft with a pre-meshed lattice architecture. Provides a scaffold for host cell proliferation and graft repopulation.
- —Pre-meshed — no intraoperative meshing equipment needed
- —Expands up to 150–170% of original size
- —Lattice design supports fluid and exudate drainage
- —APMA Seal of Approval
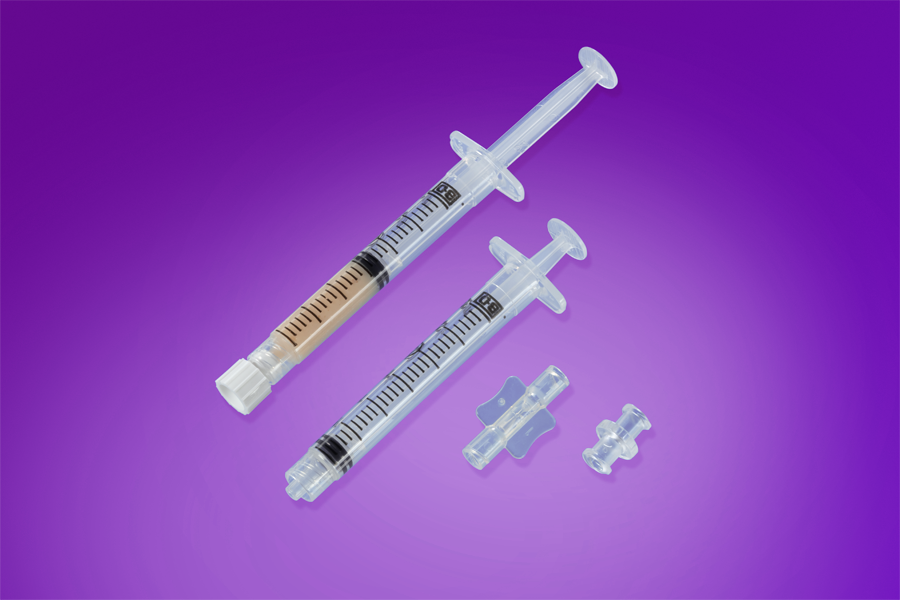

Adipose Matrix
Leneva®
Human allograft adipose tissue processed to preserve Collagen IV and Collagen VI. Functions as a scaffold for host cell infiltration and new adipocyte formation.
- —Injectable — available in 1.5cc and 3cc volumes
- —Diabetic foot ulcers, pressure ulcers, tunneling wounds
- —Fat pad reconstruction
- —APMA Seal of Approval


Placental Membrane
Salera®
Minimally processed dehydrated amnion chorion allograft that retains the structural properties of the extracellular matrix. Serves as a wound covering.
- —Bi-layer amnion and chorion membrane
- —Can be applied hydrated or dehydrated
- —3-year shelf life at ambient temperature
- —9 size configurations from 2×2cm to 7×7cm


Placental Membrane
Salera® Mini
Dehydrated amnion chorion allograft sized and packaged in milligram weight configurations for smaller wound applications or irregularly shaped wounds.
- —Dosing by weight (40mg, 80mg, 160mg)
- —Designed for smaller or tunneling wounds
- —Same 3-year ambient-temperature shelf life


Placental Matrix
AmnioBand® Membrane
Minimally processed human amnion and chorion membrane allograft. Aseptic processing preserves growth factors and matrix proteins.
- —Dehydrated bi-layer amnion and chorion
- —Can be applied hydrated or dehydrated
- —13 size configurations
- —3-year shelf life at ambient temperature
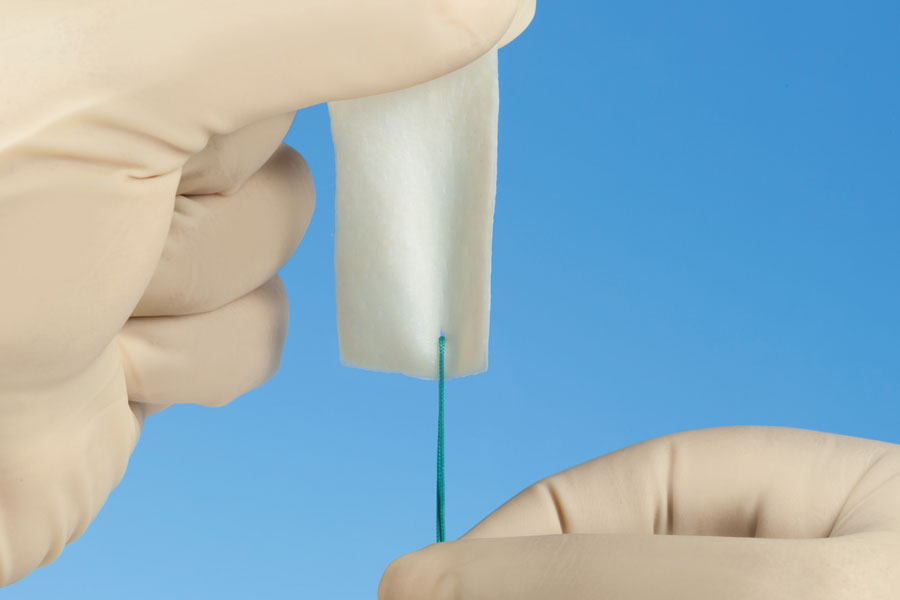

Dermal Tissue
AlloPatch® Pliable
Acellular human reticular dermal allograft, minimally processed and packaged in ethanol solution. No rehydration required.
- —Open tissue architecture for cell infiltration
- —Ready to use from package — no rehydration
- —3-year shelf life at ambient temperature
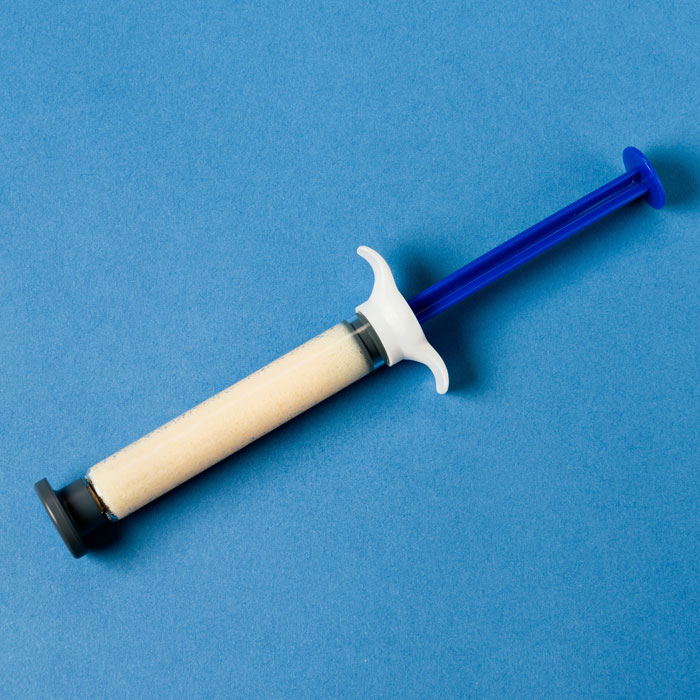

Demineralized Bone Matrix
DBX® Fiber
Fiber-form demineralized bone matrix (DBM). The latest addition to the DBX family, designed for complex anatomy and challenging fusion environments.
- —Fiber tissue form for targeted placement
- —Part of the DBX family (Putty, Mix, Fiber)
- —Spine and orthopedic applications

Viable Cellular Allograft
Trinity ELITE®
Cryopreserved viable cellular allograft containing cancellous bone and demineralized cortical bone. Intended for the treatment of musculoskeletal defects.
- —Viable cells retained within cancellous matrix
- —Available in 1.2cc, 5.3cc, 10.6cc, and 16cc
- —2-hour implantation window post-thaw
- —Processed in ISO Class 4 environment
Clinical Applications
Acute & Chronic Wounds
Acute Wounds
- ·Burns
- ·Penetrating injuries
- ·Skin cancer treatments
- ·Scar revisions
Chronic Wounds
- ·Venous ulcers
- ·Diabetic foot ulcers
- ·Pressure ulcers
- ·Vascular ulcers
Interested in MTF Biologics Wound Care?
Schedule a consultation or request product samples.
Contact Us