OSTEOCOIL
Nitinol Compression System
Sustained dynamic compression technology that harnesses the power of nitinol shape memory alloy to maintain continuous compressive force across fracture and fusion sites.
Contact Us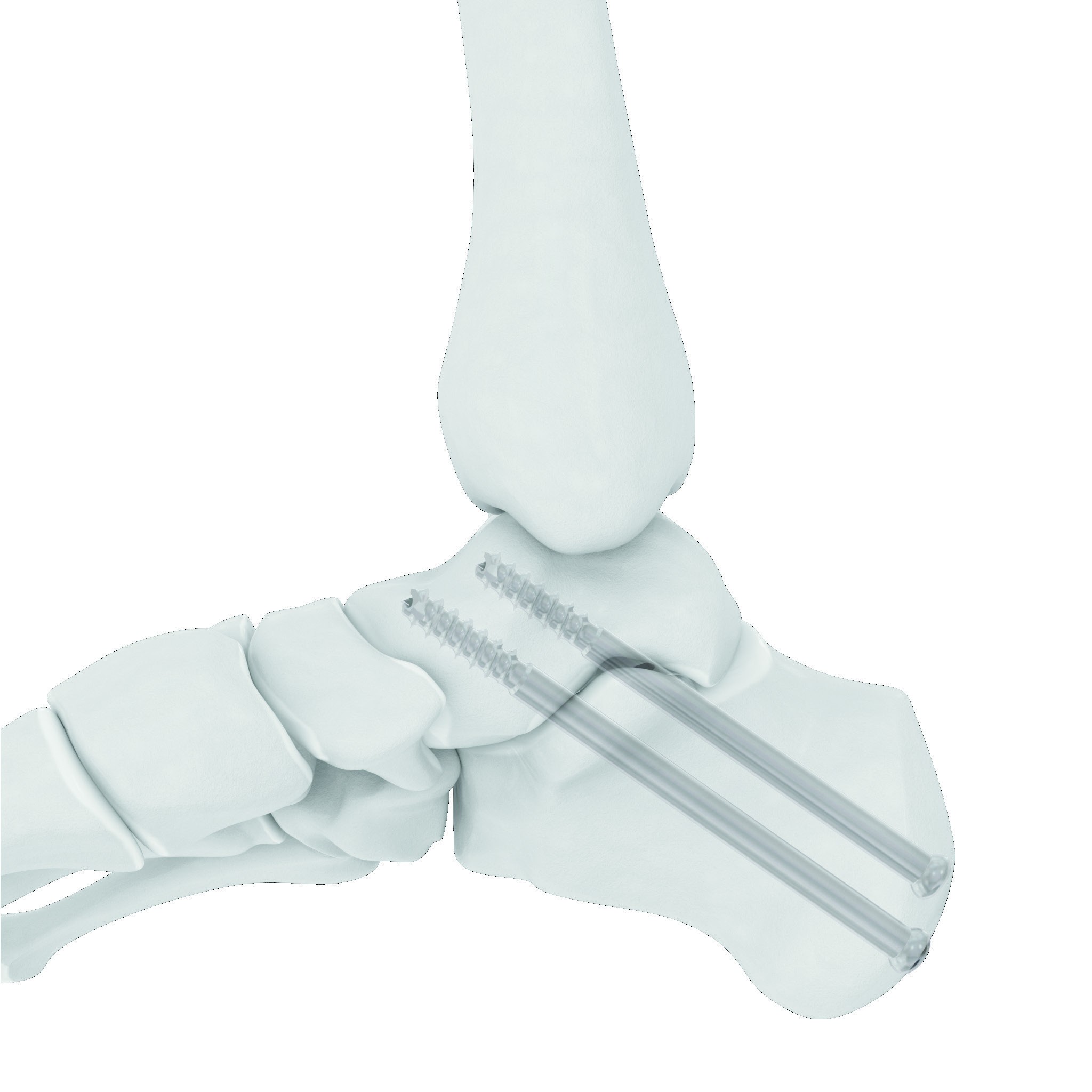
The Clinical Challenge
STANDARD IMPLANTS LOSE COMPRESSION
Conventional fixation provides an initial compression force — but that force is lost as soon as bone remodeling begins.
Compression Lost
Conventional Fixation
Typically, a compression implant provides an initial compression force which is lost as soon as there is bone length loss due to bone remodeling.
DYNAMIC
OsteoCoil Compression
The unique OsteoCoil implant results in a compression force applied across a bony fusion site even with loss of length throughout bone resorption.
HOW IT WORKS
The proprietary compressive technology allows the implant to extend as it is implanted. As the implant is inserted, the helical shaft feature expands, causing an opposing compression force between the implant head and threads.
Insertion Activates Compression
As the OsteoCoil is inserted, the proprietary helical shaft feature expands, causing an opposing compression force between the implant head and threads.
Force Applied Across the Fusion Site
The expansion of the helical shaft generates a continuous compression force across the bony fusion site — active from the moment of implantation.
Compression Maintained Through Remodeling
Unlike standard implants, OsteoCoil maintains compression even with loss of bone length throughout resorption — addressing the fundamental limitation of conventional fixation.
Precision Engineering
WHY OSTEOCOIL
The world's first and only nitinol compression system designed to provide the active biology of a staple with the placement precision of a screw.
Continuous Compression
The helical shaft feature expands during insertion, applying a compression force across the bony fusion site that persists even as bone resorption occurs.
Low-Profile Screw Heads
Designed to minimize soft-tissue irritation and reduce prominence at the implant site.
Outer Protective Sleeve
Increases torsional and bending strength, providing a robust construct during and after implantation.
FDA Cleared
510(k) cleared (K233567, November 22, 2024). Class II device indicated for small bone fusion and fracture fixation.
Documentation
Surgical Technique Guide
Surgical Technique Guide
OsteoCoil Nitinol Compression System
Interested in OsteoCoil?
Join the next generation of orthopedic surgeons utilizing dynamic compression for superior patient outcomes.
Contact Us